|
The isotope of an element is defined by the sum of the number of protons and neutrons in its nucleus. Isotope: Atoms of the same element with the same atomic number, but a different number of neutrons. Thus, each proton and neutron has a mass of about 1 amu. This isotope of carbon has 6 protons and 6 neutrons. Atomic mass is measured in Atomic Mass Units (amu), which are scaled relative to carbon, 12C, that is taken as a standard element with an atomic mass of 12. Each element is uniquely defined by its atomic number.Ītomic mass: The mass of an atom is primarily determined by the number of protons and neutrons in its nucleus. 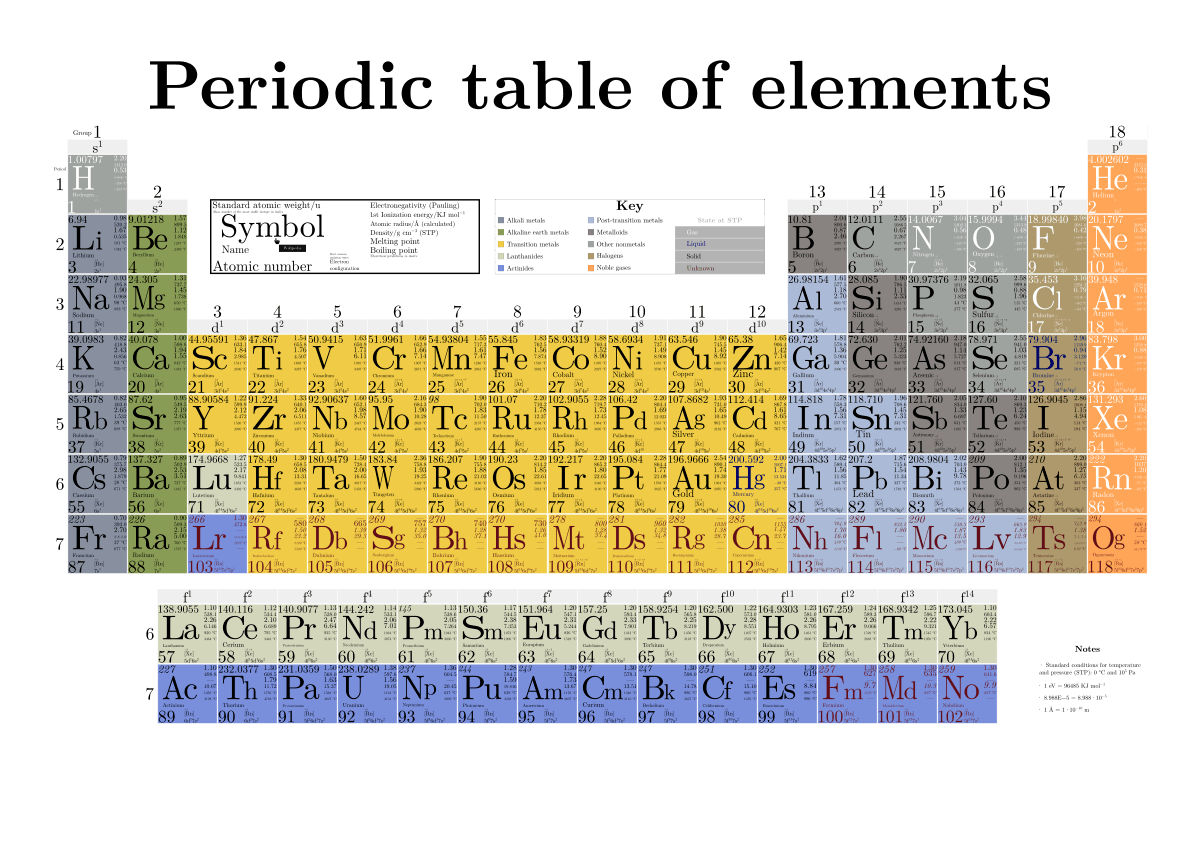
Boiling pointĪtomic number: The number of protons in an atom.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
